Are you a man 50 years or older who has been diagnosed with enlarged prostate?
If so, you may be eligible to participate in a clinical study investigating a minimally invasive method for treating benign prostatic hyperplasia (BPH).
Are urinary problems affecting your daily life? Do you find yourself…
- Having trouble starting or maintaining urination?
- Waking up several times a night to go to the bathroom?
- Feeling like your bladder isn’t empty — even after you just went?
You may be eligible to participate in a BPH Treatment Clinical Study sponsored by Olympus Corporation of the Americas. The study is evaluating two FDA approved, minimally invasive treatments for BPH — including the iTind™ procedure.

You may qualify if you:
- Are 50 years or older
- Have been diagnosed with BPH and have urinary symptoms
- Have not had prostate surgery
Participation is free and you may be compensated*
Participation includes receiving one of the treatment options being evaluated for your BPH symptoms.
There is no cost to participate in the study. Compensation may be provided for your time and/or travel during the study.
*Compensation for participating in the clinical study may vary depending on the country where the study is conducted.
Sponsored By:
Olympus Corporation of the Americas
800 West Park Drive
Westborough, MA 01581
USA
Olympus Europa SE & Co. KG
Wendenstraße 20
20097 Hamburg
Germany
What are my next steps if I want to participate?
See if you qualify. Reach out to a study site contact in your area to see if you qualify.
United States
Mayo Clinic Arizona
Study Physician:
Mitchell Humphreys
Study Contact:
Rev V Vilasan Shylaja
VilasanShylaja.Revathy@mayo.edu
480-574-1466
Arizona Urology Specialists
Study Physician:
Kalpesh Patel
Study Contact:
Jessica Castelleno
jcastellona@arizonauro.com
520-784-7042
Urology Associates of CentralCalifornia (UACC)
Study Physician:
William Schiff
Study Contact:
Kristin Santeufemia
ksanteufemia@urologyassociates.net
559-321-2924
Golden State Urology
Study Physician
Prithipal Sethi
Study Contact:
Sindhu Mohan
smohan@goldenstateurology.com
916-245-8888 ext. 308
Southeast Louisiana Veterans
Health Care System (SLVHCS)
Study Physician:
Louis Krane
Study Contact:
Kristen Lingle
Kristen.lingle@va.gov
504-905-9974
Minnesota Urology
Study Physician:
Spencer Hart
Study Contact:
Carrie McKenzie
cmckenzie@mnurology.com
651-999-6903
Adult & Pediatric Urology
Study Physician:
Sida Niu
Study Contact:
Amy Nelson
Research@adultpediatricuro.com
402-399-7894
The Feinstein Institutes for
Medical Research, Northwell Health
Study Physician:
Bilal Chughtai
Study Contact:
Alia Codelia-Anjum
acodeliaanjum1@northwell.edu
516-760-3980
Pacific West Urology
Study Physician:
Joel Abbott
Study Contact: John Dull
john.dull@pacificwesturology.com
725-225-5575
The Conrad Pearson Clinic
Study Physician:
Adam Stewart
Study Contact:
Jessica Parks
JLParks@conradpearson.com
901-464-2711
Midtown Urology Associates
Study Physician:
Tyler McClintock
Study Contact:
Andrew Barger
APBarger@cimplify.net
832-289-2585
Houston Metro Urology
Study Physician:
Mark Sutton
Study Contact:
Sarah George
sarah.george@hmutx.com
713-351-5086
The Urology Place
Study Physician:
Naveen Kella
Study Contact:
Raji Thiruppathiraj
raji@theupi.com
210-617-3670
Potomac Urology Center
Study Physician:
Nilay Gandhi
Study Contact:
Chamin Lim
clim@potomacurology.com
703-680-2111 ext. 423
United Kingdom
Queen Margaret Hospital (FIFE Health Board)
Study Physician:
Feras Al Jaafari
Study Contact:
Keith Boath
Keith.boath@nhs.scot
01383-623623 ext. 20951
Frimley Park Hospital
Study Physician:
Neil Barber
Study Contact:
Alexandra Edwards
alexandra.edwards3@nhs.net
0300-613-6779 ext. 2467
Norfolk & Norwich University Hospital
Study Physician:
Mark Rochester
Study Contact:
Claire Hayes
claire.hayes@nnuh.nhs.uk
01603-286-207 ext. 2706
A participating physician will review your medical history to ensure you meet all study participation criteria. You will also need to complete required screening tests and procedures. Following treatment, you must be available for follow-up for 5 years and should comply with your physician's instructions.
Participation is completely voluntary. All study-related procedures and treatments will be clearly explained so you can make the best decision for your health.
Impact of BPH on quality of life
Enlarged prostate symptoms, also known as BPH, impact 90% of men in their lifetime.1
If you are experiencing some or all of these symptoms, you may have BPH:
- Frequent urination or UTIs
- Painful or difficulty urinating
- Interrupted sleep
- Unable to completely empty your bladder
- Leaking
BPH is a non-cancerous condition in men where the prostate is enlarged and presses on the urethra*, causing chronic and troublesome urinary tract symptoms that can severely affect your quality of life. It’s a common condition that affects millions of men around the world.
*The urethra is a vessel which carries urine from the bladder outside the body and runs through the prostate gland.
The iTind™ procedure
The iTind procedure is a less invasive alternative to daily BPH pills, that delivers rapid, effective, and durable relief from enlarged prostate symptoms while preserving sexual function and urinary continence.2,3,4
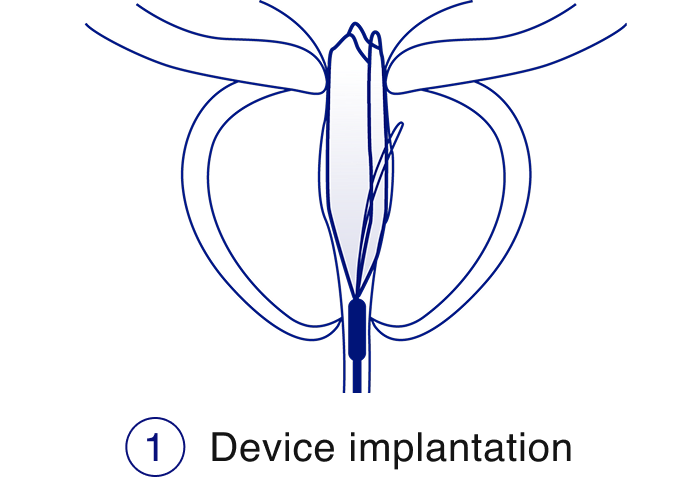
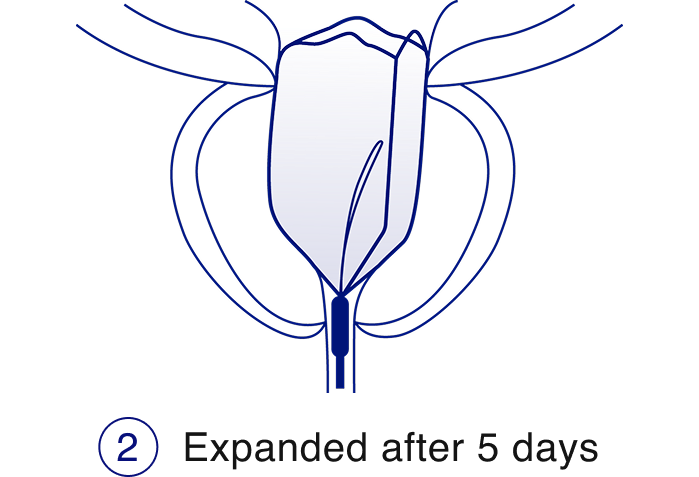
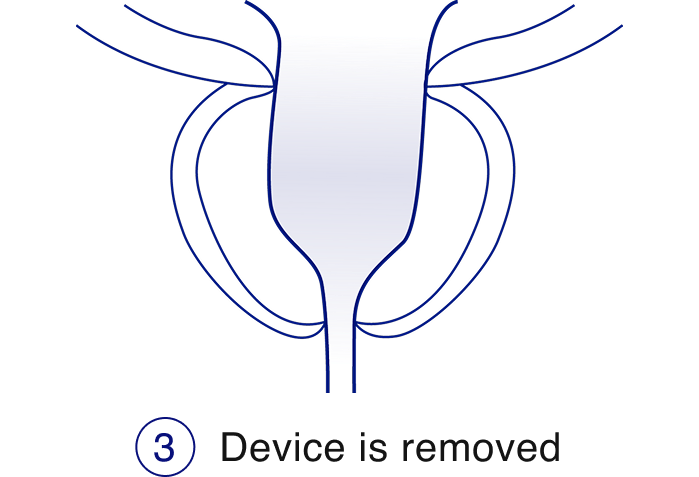
Learn more about the iTind procedure, the key benefits, and download resources.
The iTind™ procedure is intended for the treatment of symptoms due to urinary outflow obstruction secondary to benign prostatic hyperplasia (BPH) in men age 50 and above.
It is important to note that results from the iTind procedure may vary and not all patients see benefit from this treatment. As with all medical procedures results may vary. Patients should consult with their physician to determine their course of treatment.
Implantation of the iTind device may cause pelvic discomfort, blood in urine, painful or urgent urination. In rare cases, the iTind device may cause urinary tract infection or a sudden difficulty to urinate.
- Urology Care Foundation. Benign Prostatic Hyperplasia (BPH) Patient Guide. 2023.
- Chughtai B, Elterman D, Shore N, et al. The iTind Temporarily Implanted Nitinol Device for the Treatment of Lower Urinary Tract Symptoms Secondary to Benign Prostatic Hyperplasia: A Multicenter, Randomized, Controlled Trial. Urology. 2021;153:270-276. doi:10.1016/j.urology.2020.12.022.
- De Nunzio C, Cantiello F, Fiori C, et al. Urinary and sexual function after treatment with temporary implantable nitinol device (iTind) in men with LUTS: 6-month interim results of the MT-06-study. World J Urol. 2021;39(6):2037-2042. doi:10.1007/s00345-020-03418-2
- Amparore D, De Cillis S, Schulman C, Kadner G, Fiori C, Porpiglia F. Temporary implantable nitinol device for benign prostatic hyperplasia-related lower urinary tract symptoms: over 48-month results [published online ahead of print, 2023 Jun 23]. Minerva Urol Nephrol. 2023;10.23736/S2724-6051.23.05322-3. doi:10.23736/S2724-6051.23.05322-3


